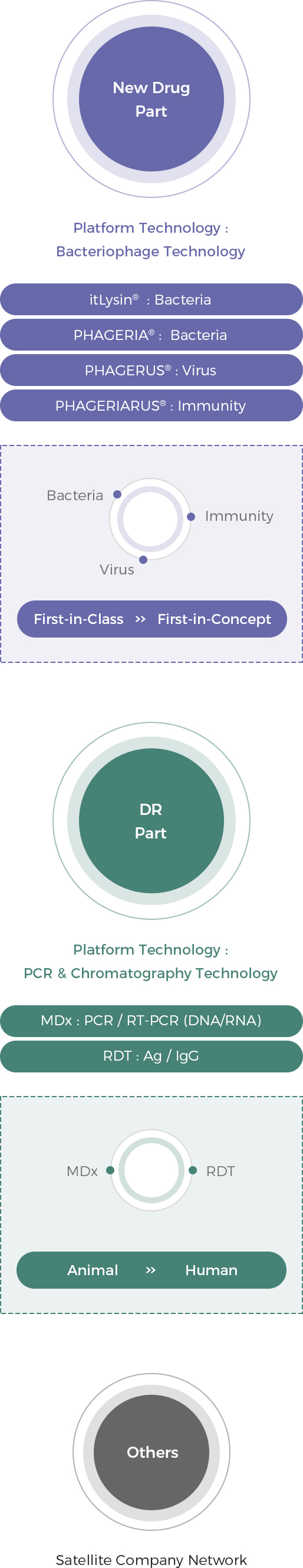
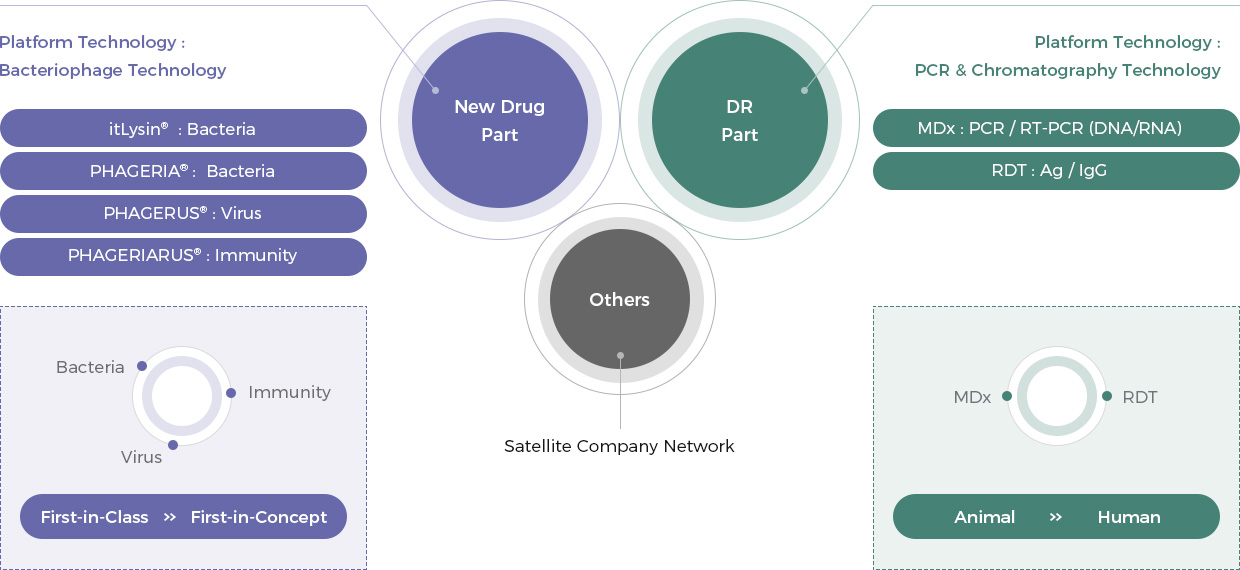
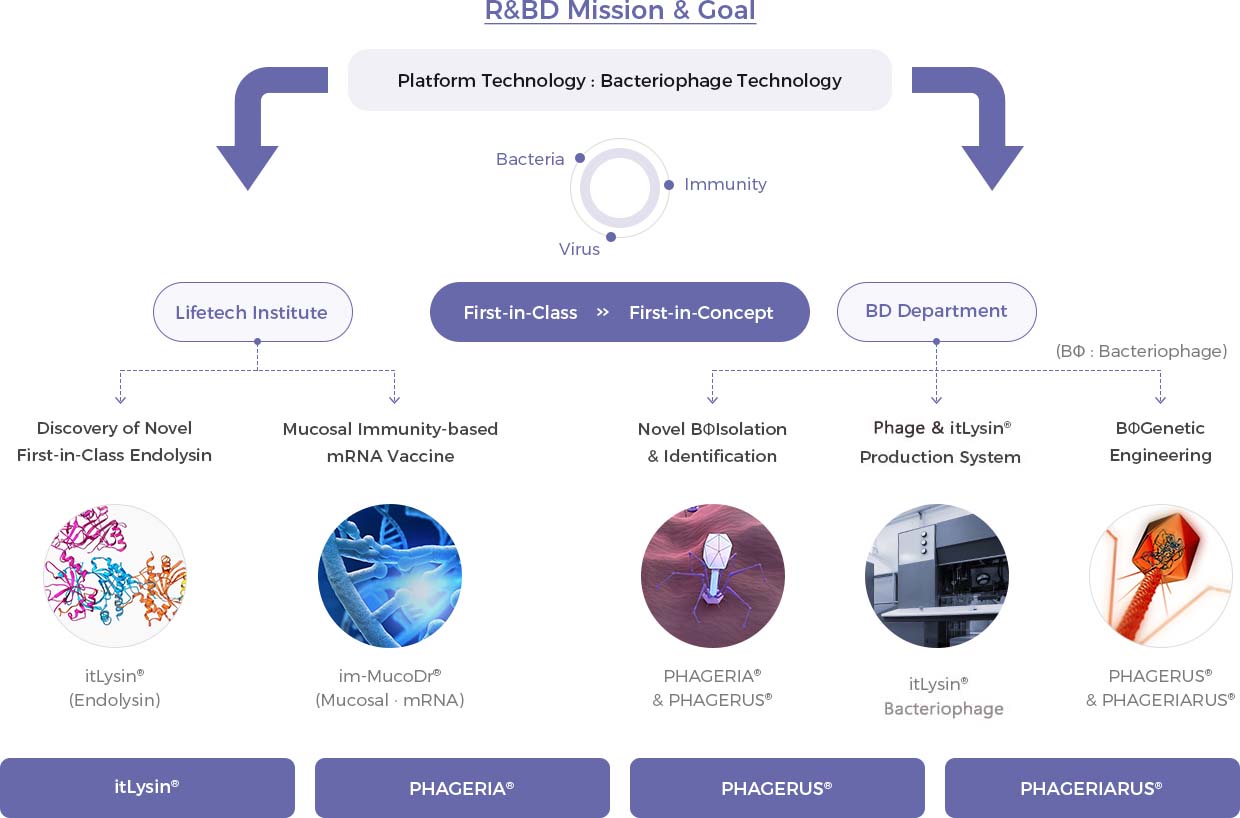
Rooted in “bacteriophage platform technology”, New Drug Part is divided into itLysin® (Endolysin) technology, PHAGERIA® technology, and PHAGERIARUS® technology. The part is focusing on developing first-in-concept drugs as well as first-in-class drugs, and it has a potential to generate mega profits in the future.
The DR Part based on “PCR & chromatography platform technology” is divided into molecular diagnosis technology and rapid IgG (Ag) diagnosis technology. The part is focusing on developing diagnostic kits for various infectious diseases, and it is generating out current revenue.
Based on the technologies derived during new drug development, we are expanding our business to the overall field of biotechnology through active partnerships with production or sales/marketing companies. We are strengthening cooperation and with our partners in a more stable and close relationship through investment in our partners.
iNtRON Biotechnology is committed to the development of innovative solutions focused on where the greatest unmet marked needs exist and our “innovative innovation” can be turned into real value for patients.
Continuous exploration in novel technologies have elevated iNtRON to the leading position in the infectious disease field based on our bacteriophage and endolysin-based, first-in-class, therapeutic agents. iNtRON has recently expanded its focus area to “immune & immunotherapeutics” beyond antibacterials.
Our passion for synthesizing “innovative innovation” does not limit us to solely therapeutics; instead, we aim to become a “Global R&BD group” where diagnostics and prophylactics are systematically co-exist as our main business and cover the entire chain systems of human health.
iNtRON believes that continuous cash-inflow is imperative for a company to maintain its sustainability. Our cash-inflow business model consists of ‘Diagnosis Biz’ and ‘Antibiotics Alternative Biz’ as a pillars for the company. iNtRON is pursuing the development of diagnostic products that can become a standard for human and animal disease diagnostics including COVID-19, Swine flu, Avian Influenza, VRE superbugs, etc. In our “Antibiotics Alternative Biz” sector, we have been making sales from our antibiotic alternative products for animals in local and overseas markets.
We are committed to becoming a ‘Global R&BD Company’. iNtRON has setup its main business model by out-licensing its mode of action (MoA) of its first-in-class pipelines to global pharmaceutical companies. iNtRON will continuously establish ‘Cash-inflow Biz’ for a more sustainable operation and most importantly, to support our passion for “innovative innovation”.
iNtRON has been focusing on the development of novel & first-in-class new drugs. To achieve this, aggressive investment in state-of-the-art research facilities and competent researchers are essential, and made possible through our out-licensing strategy business model. We have been out-licensing our novel pipelines in the early stages considering iNtRON is an R&BD focused company with full of passion for innovation.
Through the cash-inflows from out-licensing deals, iNtRON has created and established new platform technologies. This virtuous cycle will advance and lead us to becoming the “Global R&BD Group” of today.
In November of 2018, iNtRON entered into a global licensing agreement with ROIVANT Sciences for SAL200.
(https://roivant.com/roivant-sciences-and-intron-bio-sign-licensing-deal-for-novel-anti-superbugs-biologic-sal200.html)
SAL200, the leading pipeline of iNtRON, is a novel endolysin-based drug for the treatment of bacteremia caused by Staphylococcus aureus, including MRSA. After taking over the rights of SAL200, ROIVANT has been making further developments and will initiate its phase 2b study in US soon.
| Oct., 2010 | Mastitis | L/O to "C" company |
|---|---|---|
| Nov., 2018 | SAL200 | L/O to "R" company |
| Sep., 2019 | eHeme | L/O to "E" company |
| Pyocin | L/O to "B" company | |
| Jun., 2021 | iN-EYD | L/O to "R" company |
New Drug development is a field that requires a time and patience until it reaches success. For long-term R&BD, it was necessary to have a separate revenue model other than out-licensing that could provide cash-inflow on a regular basis and can cover the R&BD funds for our diverse pipelines. Since its foundation, we have setup our diagnostics development and sales business to support our researching programs. By utilizing our expertise in bacteriophage controls, we have commercialized the world’s first ‘antibiotic alternatives’ for animals based on bacteriophage.
Since our foundation, we have made continuous growth in sales of diagnostic products including PCR kits for swine flu, food poisoning, and avian influenza. Since the COVID-19 pandemic, sales and profits in our diagnostic business sector have rapidly increased, and it has grown as the major cash-cow business for iNtRON. We are currently strengthening our overseas network built by COVID-19 diagnostic kit exports to new partners. We are also developing new diagnostics products for humans (respirator, gastric, and fever & tropical diseases), animals (avian, bovine, porcine, canine, feline, and zoonosis) and food safety (halal and food poisoning).
- Possess key diagnostic MDx and RDT technology and diagnostic product design platform technology
- 35 products (as of 2020)
- 9 products to be licensed (2021~)
- With the spread of COVID-19, we experienced an increase in sales and profits in diagnostic business sector
Starting with related laws in Europe, new regulations by the U.S. Food and Drug Administration (FDA) have banned the use of antibiotics as feed supplements that help livestock and poultry grow faster in 2017. The new rules, prohibiting the over-the-counter sale to farmers of antimicrobial drugs, were enacted in an effort to inhibit the growing resistance of antibiotics in bacteria. Since then, antibiotics used as supplements in animals have been phasing out, resulting in an increased need for antibiotic alternatives. Based on our bacteriophage-based technology accumulated since our foundation, iNtRON successfully developed and commercialized the worlds first bacteriophage-based antibiotic alternatives, Our sales have continuously increased since the product launch and became one of the important cash-cow businesses for the company. iNtRON is expanding the product lineup for different animal species and infections. As we are getting additional item licenses and approvals in different countries, a rapid increase in sales is expected in coming years.
- Bacteriophage as an alternative to antibiotics for animals
- Possess highly concentrated phage fermentation technology
- Supply chain expansion based on overseas licensing with business partners
- Step-by step industrialization expansion into various fields including industrial animals
